Sorting Materials into Groups Class 6 Science NCERT Chapter 4

Key Features of NCERT Material for Class 6 Science Chapter 4 – Sorting Materials into Groups
In the previous chapter: Fibre to fabric, you studied what are fibres and the process of making fabric out of them. In chapter 4 of NCERT class 6 Science: Sorting Materials into Groups, you will study the properties of materials that things are made up of. You will also learn the classification of these materials according to their properties.
Quick revision notes
There is a tremendous assortment of objects all over. All objects around us are composed of at least one material.
A thing can be made of various materials. Numerous things can be produced using a similar material.
Classification:
The way toward arranging and gathering things as indicated by some premise is called classification.
The premise of collection:
We can aggregate materials based on the likenesses and contrasts in their properties. Every material has its own properties, that may vary from others. Nonetheless, a few properties might be normal to at least one materials.
Utilization of a material:
Uses of a material rely upon its properties and the reason for which it must be utilized.
Metals have gloss while non-metals, for the most part, have no lustre.
Smooth and rough surface: Some materials feel smooth when contacted e.g., mirror and things made of metals; though, some different materials feel rough when contacted; for instance, a bit of stone.
Hardness and softness:
Materials which can be squeezed effectively are called delicate while some different materials which can’t be squeezed are called hard; for instance, cotton is delicate while wood is hard.
Solubility
Soluble substances:
Substances that get completely dissolved in water are called soluble substances.
Insoluble substances:
Substances which cant be dissolved in water are called insoluble substances.
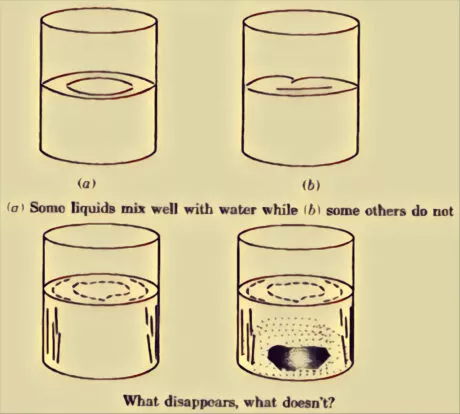
Miscible:
Those fluids which blend well in with water are supposed to be miscible.
Immiscible:
Substances which don’t blend well in with water are called immiscible.
The solubility of gases:
The solubility of gases in water is less.
Oxygen gas which is dissolved in the water is significant for the endurance of amphibian plants and creatures.
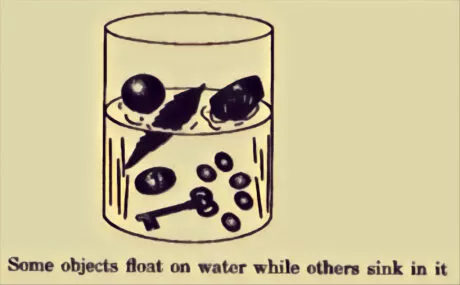
Buoyancy:
Some materials glide in the water while others sink:
Materials like sand, salt and sugar sink in water.
Materials like wax, oil and wood glide on water.
Transparency
Opaque:
Materials through which we can’t see are called opaque. For instance, wood, iron, gold.
Translucent:
Materials through which things are just somewhat noticeable are called translucent. For instance, butter paper, old glass entryway.
Transparent:
These are the materials through which things can be seen are called transparent. For instance, glass, water, air, test tube.
Things are assembled for accommodation and to contemplate their properties.
Hard: Materials which can’t be squeezed effectively are called hard e.g., stone.
Insoluble: Materials which don’t dissolve completely in the water are called insoluble.
Lustre: Some materials have an uncommon sparkle on them which is called Lustre.
Material: The matter of which an item is made, is called material. For instance, glass, steel, wood, and so on.
Metals: Materials which have certain properties like, lustre, ductility, malleability and are vibrant, good conductors of power and heat, are called metals.
Opaque: Materials which don’t permit light to go through them are called opaque.
Rough: Some materials have lopsided surface and feel rough on contacting.
Soluble: Materials which dissolve completely in the water are called soluble.
Translucent: Materials which partly permit light to go through them are called translucent.
Transparent: Materials which permit light to go through them are called transparent.
Gathering On The Basic Of Common Properties:
Objects are made of various materials. One material can be utilized to make various objects. This is conceivable in light of the fact that various kinds of materials have various properties. We need to pick materials with the correct properties dependent on what we need to utilize it for. For instance, chalk made of wood or plastic would be of no utilization since it can’t be utilized to compose on the board.
Materials have various properties like roughness, roughness, lustre, transparency, solubility, flotation, attraction towards a magnet, conduction of heat, and conduction of power. Let us study these properties individually.
Roughness:
Materials can be rough or smooth. Rough materials have bumps or edges on their surface, which can be felt by contacting them. Smooth materials do not have these bumps. Instances of rough materials are rocks, sandpaper, and bark of a tree. A glass sheet, blossom petals, and surface of an apple are a few instances of smooth surfaces (Fig. 5.1).
Luster:
Luster is the sparkle of a material. All metals in the unadulterated state are glossy and said to have shine. This property of metals is broadly utilized for making jewels and other embellishing articles. Materials like gold, silver, and bronze have radiance (Fig. 5.2).

Transparency:
 Various materials permit various measures of light to go through them relying upon a property called transparency. In view of transparency, materials can be of three sorts: Transparent, translucent, and opaque.
Various materials permit various measures of light to go through them relying upon a property called transparency. In view of transparency, materials can be of three sorts: Transparent, translucent, and opaque.
Materials that permit all the light to go through them are called transparent materials (Fig. 5.3). Glass, water, acrylic sheet, and cellophane paper are straightforward. Shopkeepers want to keep things like toffee, sweet, biscuit and so on in transparent containers with the goal that we can see them without any problem. Windows are additionally generally made of glass with the goal that light can go through and light our rooms.
Materials that permit some light to go through them are called translucent materials. Oiled paper and hued glass are translucent materials. Materials that don’t permit light to go through them are called opaque materials. Metal, leaf, stone, cardboard and wood are opaque materials.
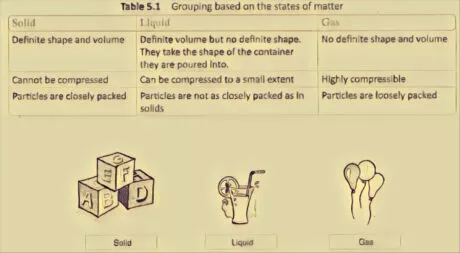
State:
All substances are comprised of matter.
Matter exists in three different states
- solid
- Liquid
- gas.
Table 5.1 Grouping dependent on the states of matter
Solubility:
Various materials have diverse solubility in water. In light of their solubility, materials can be soluble, insoluble, miscible, or immiscible.
Solid materials that dissolve completely in the water are supposed to be soluble in water. For instance, normal salt and sugar. Solid materials that don’t break down in the water are supposed to be insoluble in water. For instance, sand, wood, stone, chalk powder, and wax. Liquids that dissolve completely in the water are supposed to be miscible in water. For instance, liquor, vinegar, lemon juice, honey, and glycerine. Liquids that don’t break down in the water are supposed to be immiscible in water. For instance, kerosene, coconut oil, and diesel.
A few gases dissolve in water (e.g., carbon dioxide and oxygen). Oxygen dissolved completely in the water is fundamental for the survival of sea-going living beings. Sodas have carbon dioxide in them. Nitrogen, hydrogen, and helium are the gases which are insoluble in water.
Flotation:
Certain materials float on the water while others sink. This property of a material to glide on the water is called flotation. By and large, materials like wood, leaf, and feather float on water though rock and a metal sink.
Attraction towards a magnet:
Materials that are pulled in to a magnet are called magnetic materials. This property is magnetism. Objects made of iron are pulled in to a magnet. Alos, iron, nickel and cobalt are likewise pulled in to a magnet.
Conduction of heat:
On the off chance that you watch the utensils in your kitchen, you will see that however a large portion of them are made of metals, their handles are made of wood or hard plastic.

For what reason aren’t the handles made of metal too? This is on the grounds that metals get warmed while materials like plastic and wood don’t (Fig. 5.4). It is hard to hold the handles made of metal while cooking.
Materials that permit warmth to move through them are called conductors of heat through those that don’t permit warmth to move through them are called insulators of heat.
For the most part, metals are conductors of heat through non-metals like wood, plastic, glass, bamboo, air, and paper are insulators of warmth.
Conduction of electricity:

We get power in our homes through wires. An electric cable comprises of various metal wires with or without a plastic covering (Fig. 5.5). The metal wires lead or transmit power while the plastic covering don’t. Materials that direct power is called conductors. Materials that don’t lead power are called insulators of electricity. For instance, metals are conductors; wood, air, and plastic are insulators of electricity.
Transparent:
Materials that permit light to go through them are called transparent materials.
Translucent:
Materials that permit some light to go through them are called translucent materials.
Opaque:
Materials that don’t permit light to go through them are called opaque materials.
Miscible:
Liquids that are soluble in water are supposed to be miscible in water.
Immiscible Liquids:
Liquids that are insoluble in water are supposed to be immiscible in water.
Magnetic Materials :
They are the materials that are pulled in by a magnet.
Conductors:
Materials that conduct electricity or heath.
Insulators:
These are the materials that don’t directly heat or electricity are called protectors.
Materials have various properties that make them valuable for making various objects.
An item can be produced using various materials.
Various objects can be produced using a similar material.