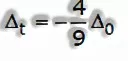Coordination Compounds: Class 12 Chemistry NCERT Chapter 9

Key Features of NCERT Material for Class 12 Chemistry Chapter 9 – Coordination Compounds
In the previous chapter 8: The D-And F-Block Elements, you learned about D-Block and F-Block elements and their workings, exceptions, etc. In this chapter: Coordination Compounds, you will learn about the gander at the significance and uses of coordination mixes. We will take a gander at the functional utilizations of these significant mixes.
Anyway, for what reason would we say we are examining such a great amount about coordination mixes? Is it just in principle or does it have any applications? All things considered, you would be astonished to know how significant these coordination mixes are, in actuality.
(Coordination Compounds: Class 12)
Review of Organic Compounds
Coordination mixes are a class of intensifies that we know as the intricate mixes. This is a direct result of the science that includes these mixes. We have thought enough about these mixes as of now, including their structures and isomers and so forth.
We realize that progress metals have this exceptional property of shaping coordination buildings. This is because of the high charge to mass proportion and furthermore the accessibility of d-orbitals. The advances in coordination science give different complex exacerbates that we use in different ventures. Coordination mixes are a typical application in different businesses. Hence, these incorporate mining and metallurgy, clinical sciences and so on to give some examples.
Quick revision notes
(Coordination Compounds: Class 12)
- Co-ordination compounds:
- A coordination compound contains a central metal atom or ion surrounded by number of oppositely charged ions or neutral molecules. These ions or molecules re bonded to the metal atom or ion by a coordinate bond.
- Example:
- They do not dissociate into simple ions when dissolved in water.
- Double salt
- When two salts in stoichiometric ratio are crystallised together from their saturated solution they are called double salts
- Example: (Mohr’s salt)
- They dissociate into simple ions when dissolved in water.
- Coordination entity:
- A coordination entity constitutes a central metal atom or ion bonded to a fixed number of ions or molecules.
- Example: In – represents coordination entity.
- Central atom or ion:
- In a coordination entity, the atom/ion to which a fixed number of ions/groups are bound in a definite geometrical arrangement around it, is called the central atom or ion.
- Example: In , is the central metal ion.
- Ligands:
- A molecule, ion or group that is bonded to the metal atom or ion in a complex or coordination compound by a coordinate bond is called ligand.
- It may be neutral, positively or negatively charged.
- Examples: etc.
- Donor atom:
- An atom of the ligand attached directly to the metal is called the donor atom.
- Example: In the complex ,CN is a donor atom.
- Coordination number:
- The coordination number (CN) of a metal ion in a complex can be defined as the number of ligand donor atoms to which the metal is directly bonded.
- Example: In the complex , the coordination number of Fe is 6.
- Coordination sphere:
- The central atom/ion and the ligands attached to it are enclosed in square bracket and are collectively termed as the coordination sphere.
- Example: In the complex is the coordination sphere.
- Counter ions:
- The ions present outside the coordination sphere are called counter ions.
- Example: In the complex , K+ is the counter ion.
- Coordination polyhedron:
- The spatial arrangement of the ligand atoms which are directly attached to the central atom/ ion defines a coordination polyhedron about the central atom.
- The most common coordination polyhedra are octahedral, square planar and tetrahedral.
- Examples: is square planar, is tetrahedral while [Cu(NH3)6]3+ is octahedral.
- Charge on the complex ion: The charge on the complex ion is equal to the algebraic sum of the charges on all the ligands coordinated to the central metal ion.
- Denticity: The number of ligating (linking) atoms present in ligand is called denticity.
- Unidentate ligands:
- The ligands whose only one donor atom is bonded to metal atom are called unidentate ligands.
- Examples:
- Didentate ligands:
- The ligands which contain two donor atoms or ions through which they are bonded to the metal ion.
- Examples: Ethylene diamine () has two nitrogen atoms, oxalate ion has two oxygen atoms which can bind with the metal atom.
- Polydentate ligand:
- When several donor atoms are present in a single ligand, the ligand is called polydentate ligand.
- Examples: In , the ligand is said to be polydentate and Ethylenediaminetetraacetate ion is an important hexadentate ligand. It can bind through two nitrogen and four oxygen atoms to a central metal ion.
- Chelate:
- An inorganic metal complex in which there is a close ring of atoms caused by attachment of a ligand to a metal atom at two points.
- An example is the complex ion formed between ethylene diamine and cupric ion, .
- Ambidentate ligand:
- Ligands which can ligate (link) through two different atoms present in it are called ambidentate ligand.
- Example: and . Here, can link through N as well as O while can link through S as well as N atom.
- Werner’s coordination theory:
- Werner was able to explain the nature of bonding in complexes.
- The postulates of Werner’s theory are:
a). Metal shows two various types of valencies: essential valence and optional valence.
b). The particles/bunches limited by auxiliary linkages to the metal have trademark spatial courses of action comparing to various coordination numbers.
c). Therefore, the most widely recognized mathematical shapes in coordination mixes are octahedral, square planar and tetrahedral.
- Primary valence
- This valence is normally ionisable.
- It is equal to positive charge on central metal atom.
- These valencies are satisfied by negatively charged ions.
- Example: In , the primary valency is three. It is equal to oxidation state of central metal ion.
- Secondary valence
- This valence is non – ionisable.
- The secondary valency equals the number of ligand atoms coordinated to the metal. It is also called coordination number of the metal.
- It is commonly satisfied by neutral and negatively charged, sometimes by positively charged ligands.
- Oxidation number of central atom: The oxidation number of the central atom in a complex is defined as the charge it would carry if all the ligands are removed along with the electron pairs that are shared with the central atom.
- Homoleptic complexes: Those complexes in which metal or ion is coordinate bonded to only one kind of donor atoms. For example:
- Heteroleptic complexes: Those complexes in which metal or ion is coordinate bonded to more than one kind of donor atoms. For example:
- Isomers: Two or more compounds which have same chemical formula but different arrangement of atoms are called isomers.
- Types of isomerism:
a). Linkage isomerism
b). Solvate isomerism or hydrate isomerism
c). Ionisation isomerism
d). Coordination isomerism
- Structural isomerism
- Stereoisomerism
a). Geometrical isomerism
b). Optical isomerism
- Structural isomerism:
- It emerges because of the distinction in structures of coordination mixes.
- Basic isomerism, or established isomerism, is a type of isomerism where particles with the equivalent sub-atomic equation have iotas fortified together in various requests.
- Ionisation isomerism:
- It arises when the counter ion in a complex salt is itself a potential ligand and can displace a ligand which can then become the counter ion.
- Example:
- Solvate isomerism:
- It is isomerism in which solvent is involved as ligand.
- If solvent is water it is called hydrate isomerism, e.g., and .
- Linkage isomerism:
- It arises in a coordination compound containing ambidentate ligand.
- In the isomerism, a ligand can form linkage with metal through different atoms.
- Example: and .
- This type of isomerism arises from the interchange of ligands between cationic and anionic entities of different metal ions present in a complex.
- Example: and .
- Stereoisomerism: This type of isomerism arises because of different spatial arrangement.
- Geometrical isomerism: It arises in heteroleptic complexes due to different possible geometrical arrangements of ligands.
- Optical isomerism: Optical isomers are those isomers which are non-superimposable mirror images.
- Valence bond theory:
- According to this theory, the metal atom or ion under the influence of ligands can use its (n-1)d, ns, np or ns, np, nd orbitals for hybridisation to yield a set of equivalent orbitals of definite geometry such as octahedral, tetrahedral, and square planar.
- These hybridised orbitals are allowed to overlap with ligand orbitals that can donate electron pairs for bonding.

- Magnetic properties of coordination compounds:
Hence, a coordination compound is paramagnetic in nature in the event that it has unpaired electrons and diamagnetic if all the electrons in the coordination compound are combined.Magnetic moment where n is number of unpaired electrons.
![]()
- Crystal Field Theory:
- It assumes the ligands to be point charges and there is electrostatic force of attraction between ligands and metal atom or ion.
- It is theoretical assumption.
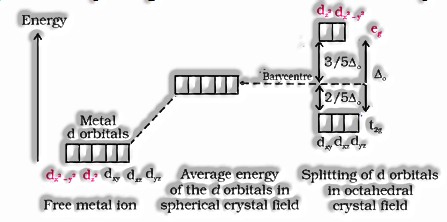
- Crystal field splitting in octahedral coordination complexes:

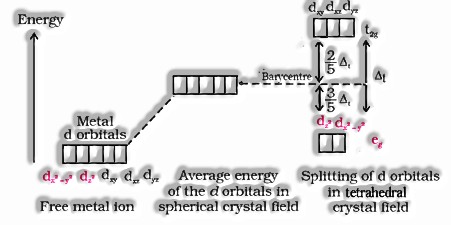
- Crystal field splitting in tetrahedral coordination complexes:

- For the same metal, the same ligands and metal-ligand distances, the difference in energy between eg and t2g level is

- Metal carbonyls:
- Metal carbonyls are homoleptic complexes in which carbon monoxide (CO) acts as the ligand.
- Example:
- The metal-carbon bond in metal carbonyls possess both s and p character.
- The M–C bond is formed by the donation of lone pair of electrons from the carbonyl carbon into a vacant orbital of the metal.
- The M–C bond is formed by the donation of a pair of electrons from a filled d orbital of metal into the vacant antibonding orbital of carbon monoxide.
- The metal to ligand bonding creates a synergic effect which strengthens the bond between CO and the metal.
(Coordination Compounds: Class 12)
Question
Q: Why do we use coordination mixes to isolate metals in extractive metallurgy?
Ans: Hence, we by and large utilize these mixes in the partition of metals during the procedure of extractive metallurgy. Therefore, this is on the grounds that these unpredictable particles have this particular property of specific precipitation and dissolvability.
(Coordination Compounds: Class 12)